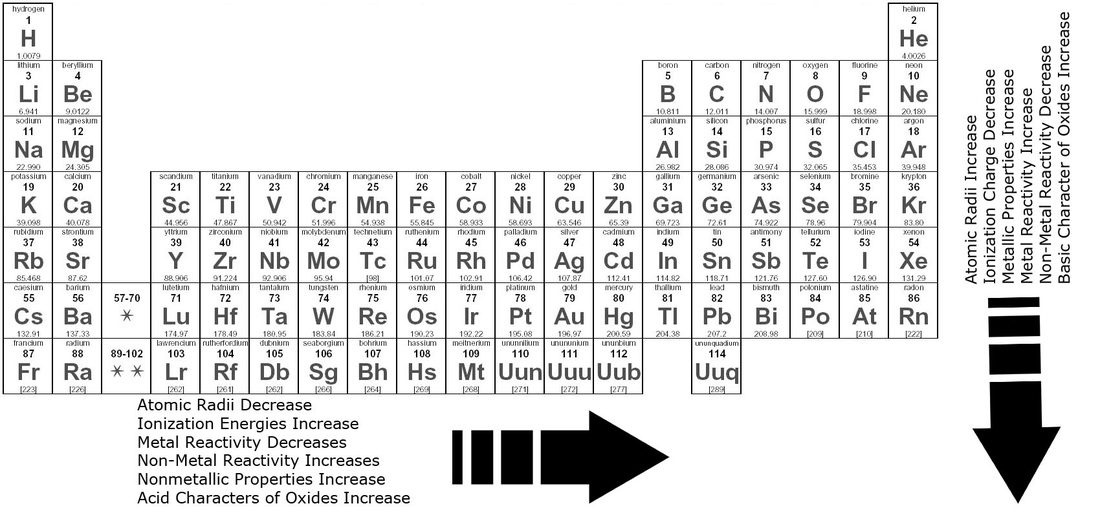
Similarities and Trends in the Periodic Table
There are certain trends and patterns in the way elements react and behave. For example the format of the periodic table is designed so properties can be easily compared. The atomic number of the elements on the periodic table are organized chronologically, starting with Hydrogen with the the atomic number of 1, going from left to right. The atomic number is the number of protons in the nucleus of an atom, therefore it is the same to the charge number of the element. The main properties that can be compared is the melting point, ionization energy, atomic radius, chemical reactivity, ion charge and conductivity.
A very reactive element means it may very vigorously react with moisture or air, but an element that is unreactive may not form any compounds at all. When elements bond with other elements, they either gain or lose an electron. The amount of energy needed to remove an electron from an atom is called the ionization energy. The ionization energy will decrease as the atomic number increases. If we magnify the elements as they bond with each other, we would be able to see the exchanging of electrons. As we look even closer, it will become noticeable that the distance between the nucleus and the electrons of an element are different. This is what is called the atomic radii; the distance between the nucleus and the electrons (trend can be seen in the image above).
Conductivity
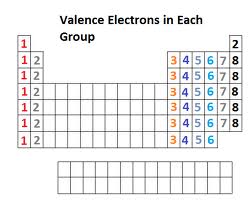
For most people, it is common knowledge that metal is good at conducting electricity. But why? As the metallic nature of an element increases, the number of valance electrons decrease. The more loose the valance electrons are, the easier it is for other electrons to jump from one atom to another, therefor being more conductive. As the temperature of a metallic conductor is lowered, the more conductive it becomes. Common conductors, such as copper and silver, have low electrical resistivity but still is limited by impurities and other defects. Even if the conductor was to be cooled to near absolute zero, it will still show some resistance.
If the temperature of a superconductor were to be cooled to under its critical temperature, then it would show no resistance. Electrons would pass through effortlessly. Superconductors were first discovered by a Dutch Physicist in 1911.
If the temperature of a superconductor were to be cooled to under its critical temperature, then it would show no resistance. Electrons would pass through effortlessly. Superconductors were first discovered by a Dutch Physicist in 1911.
Melting Point
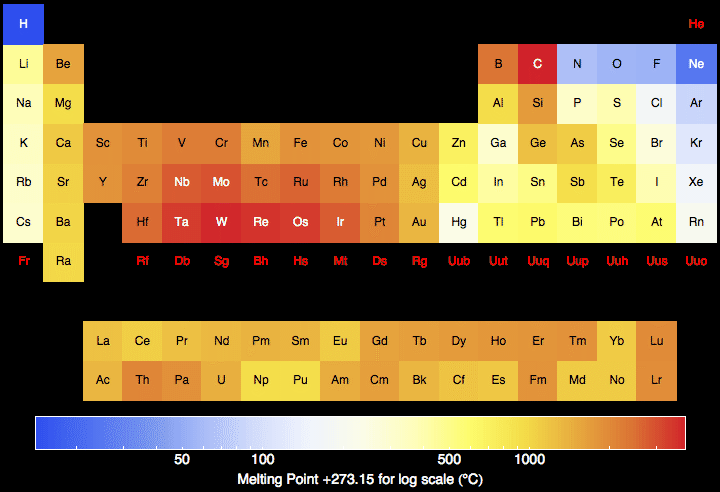
There is no obvious pattern in the melting point of the elements on the periodic table (with one end having a higher melting point and vice versa) but as expected solids have a higher melting point. Liquids and gases have a lower melting point with the exception of carbon.
The melting point of noble gases increases as their atomic number increases; meaning the atoms become heavier and denser. This trend in the melting point is the opposite for alkaline metals.
The melting point of noble gases increases as their atomic number increases; meaning the atoms become heavier and denser. This trend in the melting point is the opposite for alkaline metals.
Ion Charge
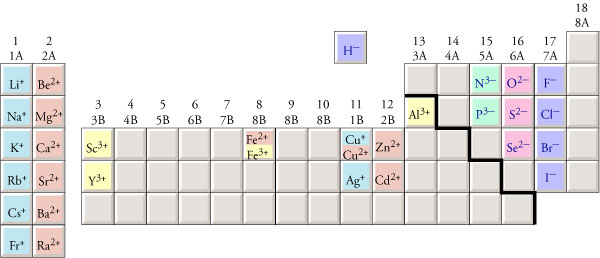
The ion charge will differ depending on which group each element belongs to. Elements from the group 1A have a charge of +1 while elements from group 2A have a charge of +2. Transition metals are in the groups with a B with a number. Group 8A are the noble gases. Because they are very stable, they do not have a charge (no valance electrons). Non-metals from 4A to 7A have a charge of -4 starting at 4A to a charge of -1, each time decreasing by -1.